What if the latest tech device had all the bells and whistles everyone wanted and was far better than anything currently being sold, but the marketer was told he could not sell it until all the previous versions of his device had broken? And then, he had to sell his fancy new device at a highly discounted price – even though its development costs had nearly bankrupted his company? That is pretty much the situation for antibiotic developers, who cannot charge enough for their drugs to support their business.
Many of the world’s giant pharmaceutical companies were either founded on or grew with the development of antibiotics, yet today these large companies for the most part have exited the field, with the majority of discovery efforts undertaken by small biotechnology companies that do not have the experience, infrastructure, or funding to support antibiotic development. This effort, which often takes a decade or longer, can cost $800M – $1B for R&D with more than 20 years before any profits are realized (see figure). As a result, in the last three years alone, after having their drugs approved following the long development process, four companies have declared bankruptcy, taking their new drugs with them.
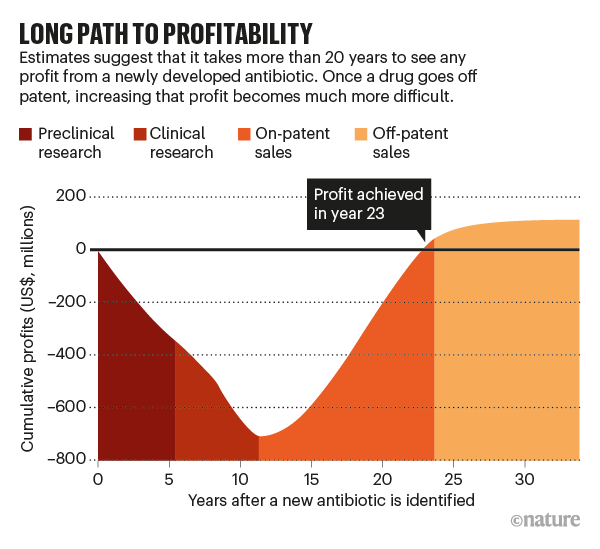
Source: Nature, Vol 584, p 338 – 341.
Though our world would be very different without antibiotics – there would be no surgeries, for example – there are several pressures that limit the use and lifespan of an antibiotic. Since bacteria will evolve to survive any threat, resistance development to new antibiotics is a major problem, particularly when these drugs are overused or used inappropriately. Antibiotic stewardship, the effort to guard against such inappropriate use by the health care system, also limits sales of newer drugs.
So while it is apparent that antibiotic resistance is a current threat, and that there is an urgent need for new drugs (Biggest Threats and Data | Antibiotic/Antimicrobial Resistance | CDC) –the low profit margins of these drugs give companies little incentive to work in the space. Why are these drugs so unprofitable?
- Their discovery is costly and lengthy and there is a high failure rate (1 in 70 are successful)
- Their prices are set low so as to be accessible and reimbursable
- Doctors only prescribe them when nothing else works to prevent resistance development
- They are taken for short periods of time, unlike other drugs, such as those for cancer, diabetes or high blood pressure (See figure)
- Resistance development results in lower antibiotic efficacy and therefore their use decreases

Source: It’s time to fix the antibiotic market (wellcome.org)
And the story is even worse for the newer, better antibiotics – these are held back from use as treatments of last resort, only after older and much cheaper drugs have failed.
There is hope, on several fronts. The AMR (Antimicrobial Resistance) Action Fund was recently started to bring two to four new antibiotics to market by 2030 with a $1B investment in smaller biotech to support clinical development. The team behind this fund includes 20 leaders in the pharmaceutical industry in collaboration with the WHO, the European Investment Bank and the Wellcome Trust (Antimicrobial Resistance Research & Development – AMR Action Fund). The US and several European countries are also exploring ways to incentivize companies to keep focus on antibiotic development. These incentives will be critical to implement given the time for research and development and the relentless emergence of new microbial threats.
You can read more here: The antibiotic paradox: why companies can’t afford to create life-saving drugs (nature.com) and here: It’s time to fix the antibiotic market (wellcome.org).




0 Comments