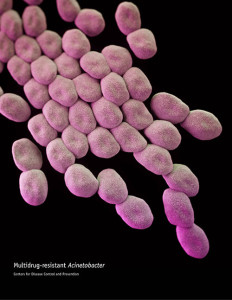
 Acinetobacter baumannii is one of the top causes of hospital-acquired infections. Most at risk for illness are the elderly and the chronically ill. Like an unwelcome guest, A. baumannii is difficult to remove once it moves into a health care setting. It has been isolated from the contaminated hands of health care workers as well as beds, sinks, countertops, door handles, computer keyboards, and blood pressure cuffs that are in the vicinity of infected patients. Acinetobacter spp can live on dry, inanimate surfaces for up to 5 months.
Acinetobacter baumannii is one of the top causes of hospital-acquired infections. Most at risk for illness are the elderly and the chronically ill. Like an unwelcome guest, A. baumannii is difficult to remove once it moves into a health care setting. It has been isolated from the contaminated hands of health care workers as well as beds, sinks, countertops, door handles, computer keyboards, and blood pressure cuffs that are in the vicinity of infected patients. Acinetobacter spp can live on dry, inanimate surfaces for up to 5 months.
Appearance:
Gram-negative rods which become spherical in the stationary phase of growth. It is catalase-positive, oxidase-negative, non-motile, and non-fermentative.
Conditions for Growth:
Aerobic. Its optimal temperature range for growth is 25° to 37°C but it can grow at 45°C. Colonies on Tryptic Soy Agar are grayish-white, circular, convex, and smooth.
Alias:
Iraqibacter. Acinetobacter baumannii caused a high number of infections among wounded American soldiers in Iraq.
Habitat:
A. baumannii has been recovered from soil and water. It also has been isolated from clinical specimens. It can colonize a patient without causing harm or, as an opportunistic pathogen, it can target moist tissues such as mucous membranes or injured skin.
Pathogenicity:
A. baumannii is a cause of hospital-acquired infections including ventilator associated pneumonia, blood stream and wound infections. Risk factors for acquiring A. baumannii include chronic illness and mechanical ventilation. According to the CDC, about 63% of Acinetobacter is considered multidrug-resistant. Although rare, this strain has been associated with community-acquired pneumonia in tropical areas like Southeast Asia.
Contamination Potential:
A. baumannii is able to adhere to environmental surfaces in the hospital and it has the ability to form biofilms on the surface of endotracheal tubes. It can be transmitted by health care personnel with colonized hands.
In 2014 the FDA recalled tattoo kits that contained ink contaminated with multiple strains of bacteria, including Acinetobacter spp.
In 2005 the FDA reported that Acinetobacter spp. and Pseudomonas spp. were isolated from media fills. The cause of the contamination was traced to cooling water which infiltrated the product container through pinhole leaks in mold plates.
Family:
Moraxellaceae, Genus: Acinetobacter.
References:
Arias, K. (2010). Contamination and Cross Contamination on Hospital Surfaces and Medical Equipment. Saxe Healthcare Communications. www.initiatives-patientsafety.org
Friedman, R. (2005). Aseptic Processing Contamination Case Studies and the Pharmaceutical
Quality System. PDA Journal of Pharmaceutical Science and Technology Vol 59; No. 2; 118-126.
Howard, A. et al., (2012) Acinetobacter baumannii: An emerging opportunistic pathogen. Virulence. May1; 3(3): 243-250
Juni, E. (2012). Genus II. Acinetobacter. In Bergey’s Manual of Systematic Bacteriology (2nd ed., Vol. 2, pp. 425-437). New York: Springer.
U.S. FDA. (August 7, 2004) FDA Warns Consumers Not to Use Certain Tattoo Inks and Tattoo Kits Sold Online. CFSAN Constituent Update.
Vaneechoutte, M. et al, (2011). Acinetobacter, Chryseobacterium, Moraxella, and Other Nonfermentative Gram-Negative Rods. In Manual of Clinical Microbiology (10th ed., Vol. 1, pp. 714-738). Washington, DC: ASM Press.



How to get rid of this dangerous bacterium?How to manage the infection caused by this bacterium?
Interested in this blog and other sources
How do I get questions and comments answered?
Hello Joseph,
Thank you for your interest in our blog. You can post your questions and comments here and we will respond, or you can always contact our Technical Support team at techsupport@microbiologics.com.