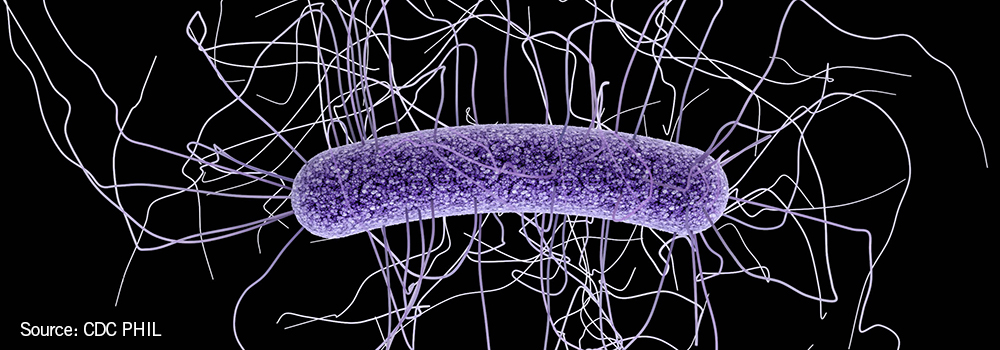
What is Clostridium difficile?
Clostridium difficile is a Gram-positive spore-forming anaerobe that can be found in the stomach and intestines of healthy people. The bacterium was first described in 1935 by Hall and O’Toole who named “difficile” because they were difficult to culture in the laboratory. There are two forms of C. difficile bacteria: an active form that cannot survive in the environment for long periods of time and a dormant form, a spore, that can survive for long periods of time. Within hours, the active form changes to the spore form in the environment such as on a toilet or door knob. Spores are very difficult to remove from surfaces and therefore can contaminate the environment by living on surfaces for weeks to months. Spores cause infection after they have been ingested and germinate into the active form of C. difficile. When the normal flora of the intestinal tract is disrupted (e.g. with antibiotics), C. difficile bacteria can multiply and produce toxins that cause mild to very severe diarrhea known as Clostridium difficile infection or CDI.
Who is at risk for C. difficile infection (CDI)?
The main risk factors for developing CDI are advanced age (>65 years) and recent antibiotic use.
What are the symptoms?
Watery liquid diarrhea lasting for two or more days is the most common symptom of CDI. Other symptoms include fever, loss of appetite, nausea, and abdominal pain/cramps.
Can an infection lead to serious illness?
C. difficile infection can lead to a more serious infection of the intestines known as pseudomembranous colitis. Pseudomembranous colitis is a condition that can lead to toxic megacolon, sepsis, and death (in rare cases). Additionally, if left untreated, serious complications of severe CDI can occur including watery diarrhea 10 to 15 times a day, blood or pus in stool, dehydration, and/or weight loss.
How is the disease transmitted?
C. difficile bacteria are found in the feces. People can become infected if they touch items or surfaces that are contaminated with feces and then touch their mouth or nose. In healthcare settings, C. difficile bacteria can be transmitted between patients on the hands of healthcare workers or through contact with contaminated items. These items can remain contaminated with C. difficile spores for days to months if not properly cleaned and disinfected.
Does the organism produce toxins? How does C. difficile damage the intestinal wall?
C. difficile bacteria cause disease by producing two large exotoxins, toxin A (an enterotoxin) and toxin B (a cytotoxin). Some C. difficile bacteria release toxin A and/or toxin B though not all strains of C. difficile bacteria produce toxin. A toxin-producing (i.e. toxigenic) strain of C. difficile bacteria must be present to cause disease. Some strains produce low levels of toxin while others are highly toxigenic. The toxins are released as C. difficile bacteria multiply in the intestine causing severe inflammation and damage to the bowel.
How is CDI diagnosed?
To determine if a person has CDI, clinical symptoms and laboratory test results should be considered. CDI is often considered in people who develop diarrhea and are at risk for CDI (e.g. recent antibiotic exposure and/or exposure to a healthcare facility within the past 3 months). A stool sample is tested by the laboratory for the presence of C. difficile and/or its toxins. In some situations, C. difficile may be diagnosed through the identification of pseudomembranes identified by endoscopy.
What laboratory tests are used to diagnose a Clostridium difficile infection?
There are several laboratory tests that can be used to detect C. difficile. A laboratory may use one or more of the following tests for a fresh stool sample: culture, antigen detection, toxin testing, and polymerase chain reaction (PCR). Culture is the most sensitive test available, but has a slow turnaround time and is labor intensive. The two most common tests used in the U.S. are enzyme immune assay for toxin A and B and PCR molecular assay. PCR is increasingly being utilized in clinical laboratories because of its high sensitivity and rapid turnaround time for results. When laboratories switch from EIA to PCR testing, they often see an increase in positive C. difficile specimens due to this increased sensitivity.
Can a patient be colonized with C. difficile but be asymptomatic? Can a colonized patient spread the disease?
Some people can be colonized, meaning their stool tests positive for C. difficile and/or its toxins (A and/or B) in the absence of clinical symptoms. Testing stool for C. difficile in a people without symptoms of CDI is generally not recommended. Additionally, no treatment is recommended for patients colonized with C. difficile.
Infected and colonized patients can shed the bacteria into the environment (e.g. toilets, rectal thermometers, etc.). C. difficile spores remain on inanimate objects indefinitely. C. difficile is easily spread within a facility when healthcare workers do not follow infection control practices, perform adequate hand hygiene, or environmental cleaning/disinfection is sub-optimal.
Have C. difficile infections become more severe or frequent in recent years? Has it become more virulent? Is it more difficult to treat?
There has been an increase in the number of reported CDI cases across the country. Also, there has been an increase in the number of severe CDI cases reported. The increase in severity is due to a more virulent strain of the bacteria called B1/NAP1.
What is C. difficile B1/NAP1/027? What is different about this strain compared to other strains? Is it epidemic in the United States?
The BI/NAP1 strain is more virulent due to increased production of toxins A and B, increased resistance to fluoroquinolones, and greater ability to form spores, which may increase its survival in the environment and transmissibility. This strain has been associated with hospital outbreaks in the U.S. and other countries. This historically uncommon strain is now epidemic throughout the U.S. Testing to identify the B1/NAP1 strain is not necessary but is used for epidemiologic purposes.
How is CDI treated?
As counterintuitive as it may seem, antibiotics are generally used to treat CDI. If a person with CDI is on antibiotics, the doctor may ask the patient to stop taking the antibiotics likely responsible for the infection and prescribe a different antibiotic to target the C. difficile bacteria. The antibiotics prescribed for the management of CDI includes metronidazole (Flagyl®) or vancomycin. CDI can be hard to treat and may recur in some individuals.
Post-treatment testing is not recommended because patients can remain colonized (unless symptoms continue). Fecal microbiota transplants, which are currently in clinical studies, are an emerging strategy for the treatment of C. difficile infections. This treatment works to restore the healthy intestinal bacteria. Research indicates this type of treatment may have a >90% success rate.
Can probiotics help treat or prevent the disease?
While there has been discussion about the use of probiotics to help treat and/or prevent CDI, currently there is insufficient evidence to support routine clinical use of probiotics to prevent or treat CDI.
How can the disease be prevented in health care settings?
CDI is a major cause of antibiotic-associated diarrhea and is the leading cause of healthcare-associated infectious diarrhea. The best transmission prevention methods are frequent hand washing and cleaning the patient environment. Extra precautions may be taken in the healthcare setting to reduce the spread of C difficile. For example, a patient with CDI may be placed in a private room with a bathroom and healthcare workers generally wear gowns and gloves while caring for the patient. Additional preventive measures include use of sporocidal or bleach-based products for environmental cleaning and a focus on early recognition of patients with CDI. Antibiotic use disrupts the normal bowel flora so judicious use of antibiotics is one of the most important keys to preventing CDI.
It’s important to remember that all healthcare workers clean their hands before entering and after leaving the patient room – whether a person has CDI or not!
Is antibiotic overuse one cause of the disease?
Antibiotics are beneficial when prescribed and taken correctly. However, improper use of antibiotics increases the chance that some organisms may adapt to the antibiotics that are designed to kill them, making the drugs less effective. Consumers should let their healthcare provider decide if antibiotics are needed to treat their illness, take the medication exactly as prescribed until it is gone, and never share antibiotics with someone they are not prescribed for. Antibiotic overuse and misuse contribute to the development and spread of CDI and other infections including those due to antibiotic resistant organisms. With few antibiotics in the pipeline, many organizations are developing programs known as Antimicrobial Stewardship Programs in effort to preserve the effectiveness of currently available antibiotics.
How does the Minnesota Department of Health know if the number of infections are increasing or decreasing?
Since 2009, the Minnesota Department of Health (MDH) has been tracking cases of CDI in select counties. Healthcare providers and laboratories in these counties report all cases of C. difficile to MDH. These data are very informative and allow MDH to determine the incidence of community- and healthcare-associated CDI, characterize the strains of reported CDI cases, and describe the epidemiology of community and healthcare-associated CDI.
What is MDH doing to prevent the spread of the disease?
CDI prevention is a priority at MDH. During 2010-2011 the Healthcare-associated Infections Unit at MDH coordinated a yearlong collaborative aimed at preventing transmission of CDI in hospitals. Eight hospitals participated in the collaborative and contributed to the development of a CDI Prevention Roadmap and Toolkit, a resource to help hospitals decrease their CDI rates. The Roadmap and Toolkit, along with other MDH CDI resources, are available on the MDH website. The collaborative was a great success due to our partnership with the Minnesota Hospital Association and the commitment and passion of hospital infection preventionists across the state to reduce the incidence and spread of CDI. MDH continues working to address CDI prevention in acute care hospitals and begin working on CDI prevention in long-term care facilities.
Looking for C. difficile controls? Visit our website to find the right strain and format in your lab. QC Sets and Panels designed specifically for instrumentation including our Cepheid C. difficile Control Panel are also available.
Biographies:
Jane Harper, RN, MS, CIC, received her Bachelor’s and Master’s degrees from the University of Michigan and is certified in infection control. She supervises the Infection Prevention and Antimicrobial Resistance Unit in the Acute Disease Investigation and Control Section at MDH and coordinates the MDH healthcare-associated infection prevention activities.
Lindsey Lesher is an epidemiologist in the Acute Disease Investigation and Control Section at MDH. She received her Bachelor’s degree at the University of California at Santa Barbara and her Master’s in Public Health at the University of Minnesota. Her current activities include MRSA surveillance and healthcare-associated infection prevention.
Kristin Shaw, MPH, CIC is an epidemiologist in the Acute Disease Investigation and Control Section at MDH. She received her Bachelor’s degree at Macalester College in St. Paul MN and her Master’s in Public Health at the University of Minnesota. Her current activities include carbapenem-resistant Enterobacteriaceae (CRE) and Clostridium difficile infection (CDI) surveillance. C. difficle QC Microorganisms



Notice must be taken in the over use of antibiotics. We must take them as prescribed by our doctor.